🌟 The AMD-11 Framework: Understanding How Age-Related Macular Degeneration Damages the Retina, and How to Protect Your Vision Proactively
Age-Related Macular Degeneration (AMD) is one of the most common causes of vision loss as we age. But AMD is not caused by a single problem. Instead, it progresses through multiple interconnected biological pathways inside the eye — involving oxidative stress, inflammation, lipid buildup, vascular changes, and genetics.
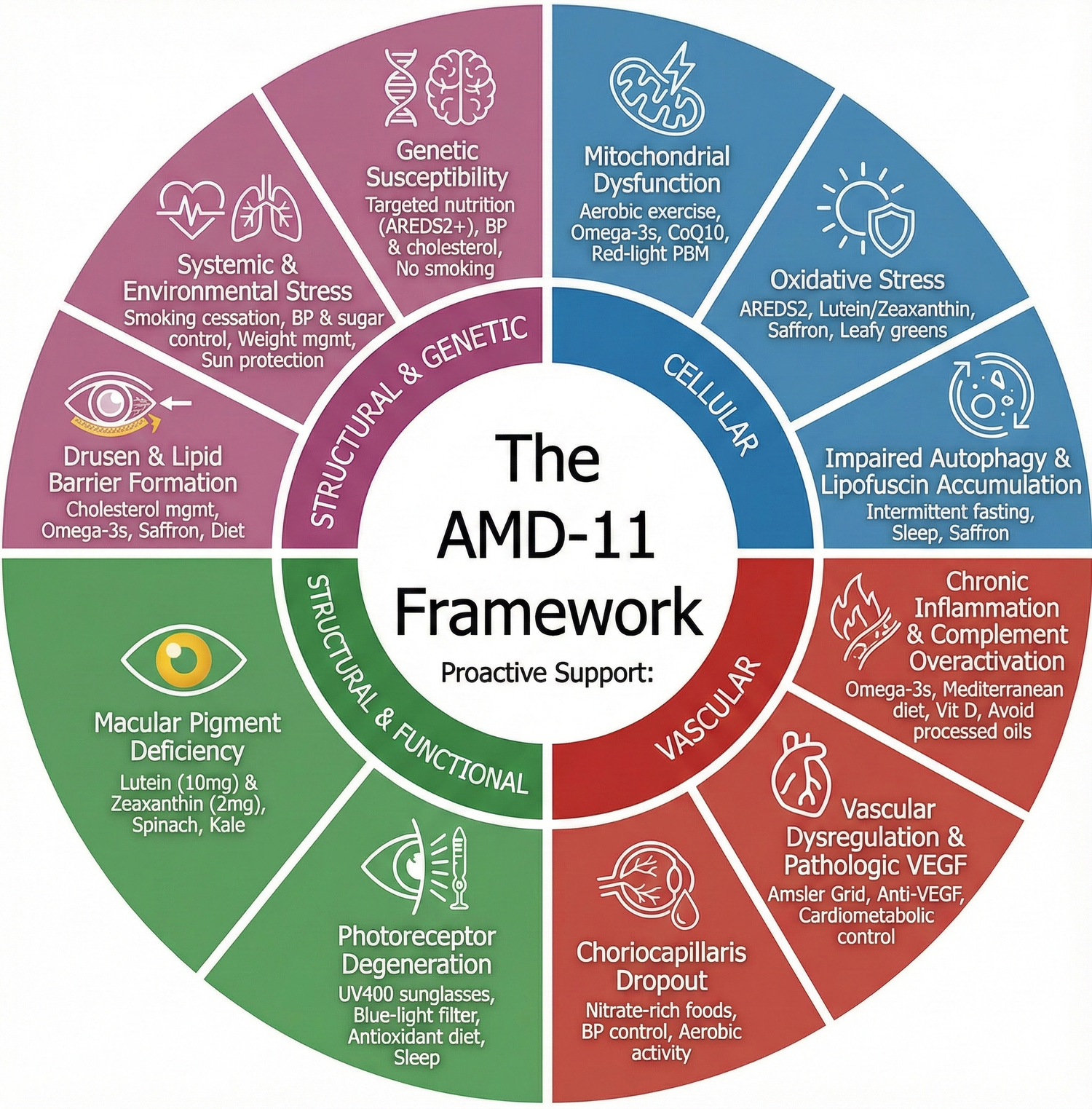
To help make this complex condition easier to understand, we’ve organized the science into a clear system called the AMD-11 Framework.
This model summarizes eleven major mechanisms behind AMD — and highlights practical steps anyone can take to support long-term macular health.
I. Cellular Mechanisms: The Retina’s “Engine Room”
The retina works extremely hard: it consumes more oxygen than nearly any other tissue in the body. That makes it vulnerable to stress at the cellular level.
1. Oxidative Stress (ROS Overload)
With age, the retina’s antioxidant defences weaken. Reactive oxygen species (ROS) build up, damaging DNA, proteins, and lipids — eventually killing retinal pigment epithelial (RPE) cells.
How to support:
- AREDS2 antioxidants (Vitamins C & E, Zinc, Copper)
- Lutein and zeaxanthin
- Dark leafy greens
- Avoid smoking
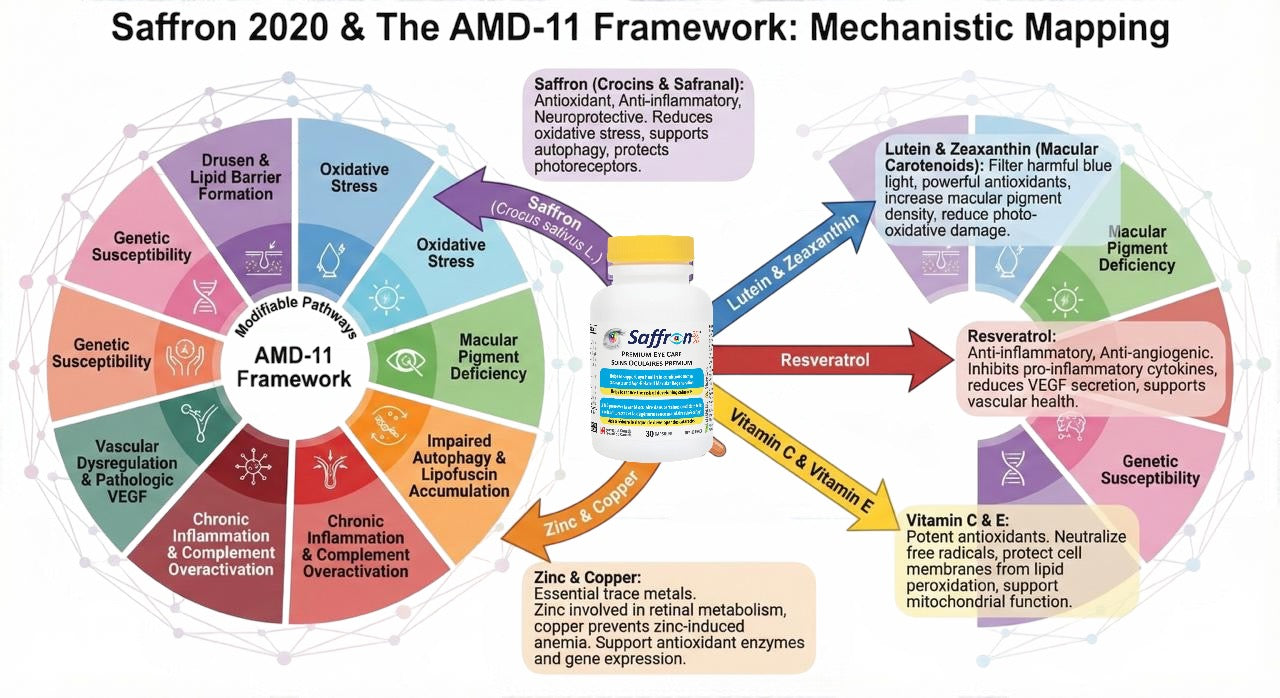
- Saffron is being studied for its ability to activate antioxidant defense genes like HO-1 and SOD
2. Mitochondrial Dysfunction (Energy Failure)
RPE cells need significant energy to recycle photoreceptor waste every day. Damaged mitochondria mean the retina can no longer keep up with this work.
How to support:
- Regular aerobic exercise
- Omega-3 fatty acids (especially DHA)
- CoQ10 and other mitochondrial-supportive nutrients
- Healthy glucose control
- Emerging interest in red-light photobiomodulation
3. Impaired Autophagy & Lipofuscin Accumulation
Autophagy is the cell’s natural “cleanup system.” When it slows down, toxic byproducts such as lipofuscin accumulate and damage the RPE.
How to support:
- Quality sleep
- Intermittent fasting or structured overnight fasts
- Better glycemic control
- Nutrition supporting cellular repair
4. Chronic Inflammation & Complement Overactivation
The complement system is part of the immune system — and in AMD, it becomes overactive. Genetic variants like CFH and ARMS2 can amplify this inflammatory response.
How to support:
- Omega-3-rich diet (salmon, sardines)
- Vitamin D sufficiency
- Mediterranean-style eating pattern
- Reducing processed omega-6 oils
II. Vascular Mechanisms: The Eye’s Oxygen Supply System
The retina relies on a dense network of tiny blood vessels, especially behind the macula in the choriocapillaris.
5. Choriocapillaris Dropout (Reduced Blood Flow)
One of the earliest AMD changes is thinning of these tiny vessels, which reduces oxygen and nutrient delivery to the macula.
How to support:
- Foods rich in natural nitrates (beets, arugula, spinach)
- Blood pressure management
- Regular physical activity
- No smoking
6. Vascular Dysregulation & VEGF Overproduction (Wet AMD)
When oxygen levels drop, the retina releases VEGF — a protein that stimulates new blood vessel growth. These vessels are often fragile and leaky, leading to bleeding and scarring.
How to support:
- Weekly Amsler grid self-checks
- Fast referral if new distortion appears
- Cardiometabolic control
- Anti-VEGF therapy for those diagnosed with wet AMD
III. Structural & Functional Changes in the Macula
These mechanisms affect the physical structure and function of the retina.
7. Drusen & Lipid Barrier Formation
Drusen are deposits made of cholesterol, lipids, complement proteins, and debris that accumulate beneath the retina. They block nutrient flow and fuel inflammation.
How to support:
- Healthy cholesterol levels (LDL control, HDL balance)
- Omega-3 intake
- Plant-rich diet
- Studies show saffron may help support lipid homeostasis in retinal cells
8. Macular Pigment Deficiency (Low Lutein & Zeaxanthin)
Macular pigment acts like natural sunglasses, filtering blue light and providing antioxidant protection. Low levels increase vulnerability to stress.
How to support:
- Supplement with lutein + zeaxanthin
- Eat spinach, kale, eggs, and corn
- Carotenoid-rich nutritional support
9. Photoreceptor Degeneration
When the supporting RPE cells fail, rods and cones lose their fuel source and begin to die, leading to blind spots and reduced clarity.
How to support:
- UV400 sunglasses
- Reduced blue-light exposure
- Consistent sleep–wake cycle
- Antioxidant-rich dietary patterns
IV. Systemic & Genetic Mechanisms
AMD risk is influenced by whole-body health.
10. Genetic Susceptibility (CFH, ARMS2, APOE)
Genetics influence complement activation, inflammation, and lipid handling.
They set the baseline risk — but lifestyle determines progression.
How to support:
- Balanced nutrition
- Optimized vitamin D
- No smoking
- Healthy blood pressure, glucose, and cholesterol
11. Systemic & Environmental Stress (Lifestyle Accelerators)
Smoking, hypertension, obesity, and high blood sugar worsen nearly every AMD pathway.
How to support:
- Smoking cessation (the #1 modifiable risk factor)
- Blood pressure control
- Stable blood sugar
- Physical activity
- Weight management
Putting the AMD-11 Framework Together
AMD is best understood not as a single disease, but as a network of interconnected stresses on the retina.
This means that even small improvements across any of the eleven pathways can contribute to better long-term macular health.
Scientific interest continues to grow around nutrition, lifestyle, and emerging natural compounds such as saffron — a key ingredient in Saffron 2020 — for their potential roles in supporting antioxidant defenses, inflammation balance, and retinal resilience.
Supporting People on Their Vision-Health Journey
At Persavita, our mission is to make complex science understandable and to empower individuals with trustworthy, evidence-based information.
The AMD-11 Framework is part of our ongoing effort to help people take proactive, meaningful steps to support macular and retinal health as they age.
Learn how Saffron 2020 protects macular health and your vision within the AMD-11 Framework by clicking here.....
References:
1. Oxidative Stress & Mitochondrial Dysfunction (Cellular)
- Mimura T. Oxidative Stress in Age-Related Macular Degeneration. 2025.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12561695/ PMC - Qu S et al. Age-Related Macular Degeneration and Mitochondria-Targeted Therapies. Int J Mol Sci. 2024.
https://www.mdpi.com/1422-0067/25/3/1624 MDPI - Tong Y et al. Role of Mitochondria in Retinal Pigment Epithelial Aging and AMD. Front Aging. 2022.
https://www.frontiersin.org/articles/10.3389/fragi.2022.926627/full Frontiers - Davinelli S et al. Sleep and Oxidative Stress: Current Perspectives on the Role of Sleep in Redox Homeostasis.Antioxidants. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11199221/ PMC - Vaccaro A et al. Sleep Loss Can Cause Death through Accumulation of Reactive Oxygen Species in the Gut. Cell. 2020.
https://www.cell.com/cell/fulltext/S0092-8674(20)30555-9 Cell
2. Autophagy, Lipofuscin & Cellular “Waste” Clearance
- Zhang Z et al. Autophagy in Dry AMD: A Promising Therapeutic Strategy for Retinal Degeneration. Prog Retin Eye Res. 2024.
https://pubmed.ncbi.nlm.nih.gov/38593971/ PubMed - Kaarniranta K et al. Autophagy in Age-Related Macular Degeneration. Autophagy. 2023.
https://www.tandfonline.com/doi/full/10.1080/15548627.2022.2069437 Taylor & Francis Online - Intartaglia D et al. Autophagy in the Retinal Pigment Epithelium: A New Vision and Future Challenges. FEBS J. 2022.
https://febs.onlinelibrary.wiley.com/doi/10.1111/febs.16018 FEBS Journal - Shahhossein-Dastjerdi S et al. Autophagy and Exocytosis of Lipofuscin in Human Retinal Pigment Epithelium.IOVS. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11044829/ PMC - Si Z et al. The Role of Retinal Pigment Epithelial Cells in AMD: Phagocytosis and Autophagy. Biomolecules. 2023.
https://www.mdpi.com/2218-273X/13/6/901 MDPI
3. Chronic Inflammation & Complement Overactivation
- Deng Y et al. Age-Related Macular Degeneration: Epidemiology, Genetics, and Pathophysiology. Cell Prolif. 2022.
https://www.sciencedirect.com/science/article/pii/S2352304221000295 ScienceDirect - Zhang Z et al. (same as #6) – links oxidative stress, lipofuscin, NLRP3 inflammasome and complement activation in AMD. PubMed
4. Choriocapillaris Dropout & Vascular Dysregulation
- Lipecz A et al. From Mechanisms of Choriocapillaris Aging to Novel Interventions. GeroScience. 2019.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6925092/ PMC - Lutty GA. Choriocapillaris Dropout in Early Age-Related Macular Degeneration. Trans Vis Sci Tech. 2020.
https://pmc.ncbi.nlm.nih.gov/articles/PMC7216757/ PMC - Neri G et al. Choriocapillaris in Age-Related Macular Degeneration. Int J Mol Sci. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11590706/ PMC - Seddon JM et al. Choroidal Vascular Loss in Age-Related Macular Degeneration. JAMA Ophthalmol. 2016.
https://jamanetwork.com/journals/jamaophthalmology/fullarticle/2553799 JAMA Network - Sohn EH et al. Choriocapillaris Degeneration in Geographic Atrophy. Am J Pathol. 2019.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6616998/ ajp.amjpathol.org
5. VEGF-Driven Neovascularization & Anti-VEGF Therapy
- Cheng S et al. Treatment of Neovascular Age-Related Macular Degeneration: Anti-VEGF Agents and Emerging Therapies. Front Med. 2024.
https://www.frontiersin.org/articles/10.3389/fmed.2024.1411278/full Frontiers - Bidiwala S et al. Efficacy of Anti-VEGF Therapy in Neovascular AMD: A Review. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11684535/ PMC - Moraru AD et al. Guideline Recommendations for Optimal Anti-VEGF Therapy in Neovascular AMD. Life. 2024.
https://www.mdpi.com/2075-1729/14/10/1220 MDPI - Dervenis N et al. Neovascular Age-Related Macular Degeneration. BMJ Open Ophthalmol. 2024.
https://bmjophth.bmj.com/content/9/1/e001516 bmjophth.bmj.com
6. Drusen, Lipids & Barrier Formation
- Ban N et al. Drusen in AMD from the Perspective of Cholesterol Metabolism. Prog Retin Eye Res. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11084323/ PMC - Apte RS et al. Targeting Tissue Lipids in Age-Related Macular Degeneration. Trends Mol Med. 2016.
https://www.sciencedirect.com/science/article/pii/S2352396416300342 ScienceDirect - Xu Q et al. Understanding AMD by Analogy: Systematic Review of Lipid-Related Extracellular Deposits. Lipids Health Dis. 2018.
https://lipidworld.biomedcentral.com/articles/10.1186/s12944-017-0647-7 SpringerLink - UMass Med – Punzo Lab. Age-Related Macular Degeneration (AMD).
https://www.umassmed.edu/punzolab/research/amd/ UMass Chan Medical School
7. Macular Pigment, Lutein/Zeaxanthin & Blue-Light Protection
- Lima VC et al. Macular Pigment in Retinal Health and Disease. Int J Retina Vitreous. 2016.
https://journalretinavitreous.biomedcentral.com/articles/10.1186/s40942-016-0044-9 SpringerLink - Whitehead AJ et al. Macular Pigment: A Review of Current Knowledge. Arch Ophthalmol. 2006.
https://jamanetwork.com/journals/jamaophthalmology/fullarticle/417803 JAMA Network - Scripsema NK et al. Lutein, Zeaxanthin, and Meso-Zeaxanthin in Clinical Practice. J Ophthalmol. 2015.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4706936/ PMC - Mrowicka M et al. Lutein and Zeaxanthin and Their Roles in AMD. Nutrients. 2022.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8874683/ PMC - National Eye Institute (NEI). AREDS/AREDS2 Clinical Trials.
https://www.nei.nih.gov/research/clinical-trials/age-related-eye-disease-studies-aredsareds2/about-areds-and-areds2 National Eye Institute - Keenan TDL et al. Oral Antioxidant and Lutein/Zeaxanthin Supplements Slow GA Progression in AMD.Ophthalmology. 2025.
https://pubmed.ncbi.nlm.nih.gov/39025435/ PubMed - Evans JR, Cochrane Review. Antioxidant Vitamin and Mineral Supplements for Slowing AMD. 2023.
https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD000254.pub5/abstract Cochrane Library
8. Photoreceptor Degeneration & Functional Loss
- Curcio CA et al. Photoreceptor Loss in Age-Related Macular Degeneration. Invest Ophthalmol Vis Sci. 1996.
https://pubmed.ncbi.nlm.nih.gov/8641827/ PubMed - Curcio CA et al. Photoreceptor Topography in Ageing and Age-Related Maculopathy. Eye. 2001.
https://www.nature.com/articles/eye2001140.pdf Nature - Jackson GR et al. Photoreceptor Degeneration and Dysfunction in Aging and ARM. Am J Ophthalmol. 2002.
https://pubmed.ncbi.nlm.nih.gov/12067593/ PubMed - Shelley EJ et al. Cone Degeneration in Aging and AMD. Arch Ophthalmol. 2009.
https://jamanetwork.com/journals/jamaophthalmology/fullarticle/422827 JAMA Network - Cheng SY et al. Altered Photoreceptor Metabolism Causes Late-Stage AMD-Like Pathology. PNAS. 2020.
https://www.pnas.org/doi/10.1073/pnas.2000339117 PNAS
9. Genetic Susceptibility (CFH, ARMS2, Complement Genes)
- Schwartz SG et al. Genetics and AMD: A Practical Review for Clinicians. Br J Ophthalmol. 2016.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4938141/ PMC - Klein R et al. Risk Alleles in CFH and ARMS2 and Long-Term Incidence of AMD. Arch Ophthalmol. 2013.
https://jamanetwork.com/journals/jamaophthalmology/fullarticle/1390428 JAMA Network - Scholl HPN et al. CFH, C3 and ARMS2 Are Significant Risk Loci for Geographic Atrophy. PLoS ONE. 2009.
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0007418 PLOS - Kumaramanickavel G et al. Age-Related Macular Degeneration: Genetics and Biology. Semin Ophthalmol. 2016.
https://www.sciencedirect.com/science/article/pii/S2162098923003080 ScienceDirect
10. Systemic & Lifestyle Risk Factors (Smoking, BP, Metabolic Health)
- Babaker R et al. Risk Factors for Age-Related Macular Degeneration: Updated Systematic Review and Meta-Analysis. Medicine (Baltimore). 2025.
https://journals.lww.com/md-journal/fulltext/2025/02210/risk_factors_for_age_related_macular_degeneration_.71.aspx Lippincott Journals - Kuan V et al. Association of Smoking, Alcohol, Blood Pressure, BMI and Glycemic Traits with AMD. JAMA Ophthalmol. 2021.
https://jamanetwork.com/journals/jamaophthalmology/fullarticle/2785704 JAMA Network - Heesterbeek TJ et al. Risk Factors for Progression of Age-Related Macular Degeneration. Ophthalmic Physiol Opt. 2020.
https://onlinelibrary.wiley.com/doi/10.1111/opo.12675 Wiley Online Library - Saigal K et al. Modifiable Lifestyle Risk Factors and Strategies for Slowing AMD. Vision. 2025.
https://www.mdpi.com/2411-5150/9/1/16 MDPI - Macular Society. Nutrition and AMD.
https://www.macularsociety.org/support/daily-life/practical-guides/healthy-living/nutrition/ Macular Society


Leave a comment
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.